
Abstract
Recently, Nanjing Liming Bio-Products Co., Ltd. (www.limingbio.com)SARS-CoV-2 lgM/IgG Antibody Rapid Test Kit has been certified by the Brazilian National Health Supervision Bureau and obtained the ANVISA certification. At the same time, the SARS-CoV-2 RT-PCR and the IgM/IgG antibody rapid test kit are also listed on the official recommended procurement list of Indonesia. Meanwhile, Liming Bio StrongStep® Novel Coronavirus (SARS-CoV-2) Multiplex Real-time PCR Kit, was approved by The Singapore Health Sciences Authority (HSA) , and obtained the HSA certificate.

Picture 1 the Brazil ANVISA certification
Brazil (ANVISA) certification
ANVISA, known as Agência Nacional de Vigilância Sanitária, is the Brazilian medical device regulator. It is necessary for a company to be registered with ANVISA, the National Health Supervision Agency, to sell medical devices legally in Brazil. To be certified, those medical devices entering Brazil must meet the requirements of Brazilian GMP along with the specific standards set by the Brazilian authorities. In Brazil, IVD medical devices are classified into Class I, II, III, and IV according to the risk level from low to high. For Class I and II products, the Cadastro approach is adopted, while for Class III and IV products, the Registro approach is utilized. After successful registration, a registration number will be issued by ANVISA, and the data will be uploaded to the Brazilian medical device database, this number and its corresponding registration information will appear on DOU (Diário Oficial da União).


Picture 2 Singapore Health Sciences Authority (HSA) certificate


Picture 3 The official recommended procurement list of Indonesia

Picture 4 StrongStep® SARS-CoV-2 IgM/IgG Antibody Rapid Test
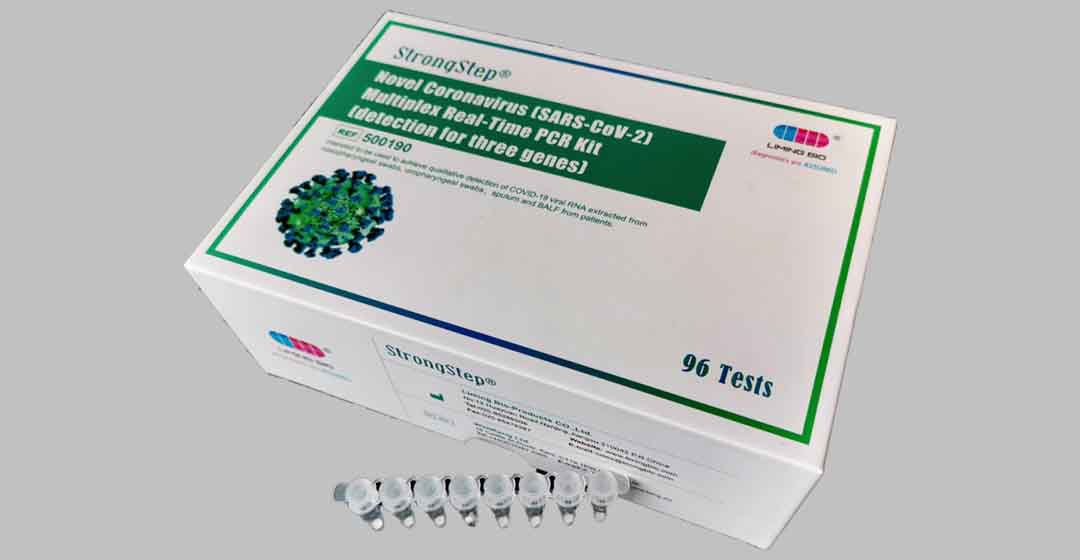
Picture 5 Novel Coronavirus (SARS-CoV-2) Multiplex Real-Time PCR kit
Note:
This highly sensitive, ready-to-use PCR kit is available in lyophilized format (freeze-drying process) for long-term storage. The kit can be transported and stored at room temperature and is stable for one year. Each tube of premix contains all of the reagents needed for the PCR amplification, including Reverse-transcriptase, Taq polymerase, primers, probes, and dNTPs substrates. It only need add 13ul distilled water and 5ul extracted RNA template , then it can be run and amplified on the PCR instruments.
The difficulty of cold chain transportation of Novel Coronavirus nucleic acid detection reagent
When conventional nucleic acid detection reagents are transported in long distance, the (-20±5) ℃ cold chain storage and transportation is required to ensure the bioactive of enzyme in the reagents remain active. To make sure that the temperature reaches the standard, several kilograms of dry ice are needed for each box of nucleic acid testing reagent even less than 50g , but it can only last for two or three days. On the perspective of industry practice, the actual weight of reagents issued by manufacturers is less than 10% (or far less than this value) of the container. Most of the weight comes from dry ice, ice packs and foam boxes, so the transportation cost is extremely high.
In March 2020, COVID-19 began to break out in a large scale abroad, and the demand for Novel Coronavirus nucleic acid detection reagent increased dramatically. Despite the high cost of exporting the reagents in the cold chain, most manufacturers can still accept it due to the large quantity and high profit.
However, with the improvement of national export policies for anti-pandemic products, as well as the upgrading of national control over the flow of people and logistics, there is extension and uncertainty in the transportation time of reagents, which resulted in prominent product problems caused by the transportation. Extended transportation time (transportation time of about half a month is very common) leads to frequent product failures when the product reaches the client. This has troubled most nucleic acid reagents export enterprises.
Lyophilized technology for PCR reagent helped the transportation of Novel Coronavirus nucleic acid detection reagent worldwide
The lyophilized PCR reagents can be transported and stored at room temperature, which can not only reduce the transportation cost, but also avoid the quality problems caused by transportation process. Therefore, lyophilizing the reagent is the best way to solve the problem of export transportation.
Lyophilization involves freezing a solution into a solid state, and then sublimate and separate the water vapor under vacuum condition. The dried solute remains in the container with the same composition and activity. Compared with conventional liquid reagents, the full-component lyophilized Novel Coronavirus nucleic acid detection reagent produced by Liming Bio has the following characteristics:
Extremely strong heat stability: it can with stand treatment at 56℃ for 60 days, and the morphology and performance of the reagent remain unchanged.
Normal temperature storage and transportation:no need for cold chain, no need to store at low temperature before unsealing, fully release the cold storage space.
Ready-to-use: lyophilizing of all components, no need for system configuration, avoiding the loss of components with high viscosity such as enzyme.
Multiplex targets in one tube: the detection target covers novel coronavirus ORF1ab gene, N gene, S gene to avoid the virus genovariation. In order to reduce false negative, human RNase P gene is used as internal control, so as to meet the clinical need for sample quality control.
SARS-CoV-2 IgM/IgG Antibody Rapid Test and Novel Coronavirus (SARS-CoV-2) Multiplex Real-Time PCR Kit (detection for three genes) has previously been CE marked in the UK, and now accepted and being processed by EUA of FDA in America.
Nanjing Liming Bio-Products Co., Ltd. has always put the quality of the test kit in the first place, and is concentrating on the expansion of the capacity. The company will provide high-quality COVID-19 testing products and services to medical institutions all around the world, and contribute to the global epidemic prevention and control, so as to build a global community of shared future.
Long Press~Scan and Follow Us
Email: sales@limingbio.com
Website: https://limingbio.com

Post time: Jul-06-2020







