COVID-19
-
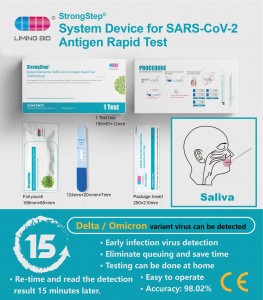
StrongStep System Device for SARS-CoV-2 Antigen Rapid Test
REF 500210 Specification 1 Test/box Detection principle Immunochromatographic assay Specimens SalivaIntended Use StrongStep System Device for SARS-CoV-2Antigen Rapid Test employs immunochromatography technobgy to detect the SARS-CoV-2 nucleocapsid antigen in human saliva. This test is single use only and intended for self-testing. It is recommended to use this test within 7 days of symptom onset. lt is supported by the dinical performance assessment. -

SARS-CoV-2 Antigen Rapid Test(nasal)
REF 500200 Specification 1 Tests/Box ;5 Tests/box ; 20 Tests/box Detection principle Immunochromatographic assay Specimens Anterior nasal swab Intended Use StrongStep® SARS-CoV-2 Antigen Rapid Test Cassette employs immunochromatography technology to detect the SARS- CoV-2 nucleocapsid antigen in human anterior nasal swab specimen. This testis single use only and intended for self-testing. It is recommanded to use this test within 5 days of symptom onset. It is supported by the clinical performance assessment. -

SARS-CoV-2 Antigen Rapid Test(Professional Use)
REF 500200 Specification 25 Tests/box Detection principle Immunochromatographic assay Specimens Anterior nasal swab Intended Use StrongStep® SARS-CoV-2 Antigen Rapid Test Cassette employs immunochromatography technology to detect the SARS- CoV-2 nucleocapsid antigen in human anterior nasal swab specimen. This testis single use only and intended for self-testing. It is recommanded to use this test within 5 days of symptom onset. It is supported by the clinical performance assessment. -
1人份抗原卡实物图唾液版1_00_副本-300x216.png)
SARS-CoV-2 Antigen Rapid Test for Saliva
REF 500230 Specification 20 Tests/Box Detection principle Immunochromatographic assay Specimens SalivaIntended Use This is a rapid immunochromatographic assay for the detection of SARS-CoV-2 virus Nucleocapsid Protein antigen in human Saliva swab collected from individuals who are suspected of COVID-19 by their healthcare provider within the first five days of the onset of symptoms. The assay is used as an aid in the diagnosis of COVID-19. -

System Device for SARS-CoV-2 & Influenza A/B Combo Antigen Rapid Test
REF 500220 Specification 20 Tests/Box Detection principle Immunochromatographic assay Specimens Nasal / Oropharyngeal swab Intended Use This is a rapid immunochromatographic assay for the detection of SARS-CoV-2 virus Nucleocapsid Protein antigen in human Nasal/Oropharyngeal swab collected from individuals who are suspected of COVID-19 by their healthcare provider within the first five days of the onset of symptoms. The assay is used as an aid in the diagnosis of COVID-19. -

Dual Biosafety System Device for SARS-CoV-2 Antigen Rapid Test
REF 500210 Specification 20 Tests/Box Detection principle Immunochromatographic assay Specimens Nasal / Oropharyngeal swab Intended Use This is a rapid immunochromatographic assay for the detection of SARS-CoV-2 virus Nucleocapsid Protein antigen in human Nasal /Oropharyngeal swab collected from individuals who are suspected of COVID-19 by their healthcare provider within the first five days of the onset of symptoms. The assay is used as an aid in the diagnosis of COVID-19. -

Novel Coronavirus (SARS-CoV-2) Multiplex Real-Time PCR Kit
REF 500190 Specification 96 Tests/Box Detection principle PCR Specimens Nasal / Nasopharyngeal swab Intended Use This is intended to be used to achieve qualitative detection of SARS-CoV-2 viral RNA extracted from nasopharyngeal swabs, oropharyngeal swabs, sputum and BALF from patients in association with an FDA/CE IVD extraction system and the designated PCR platforms listed above. The kit is intended for use by laboratory trained personnel
-

SARS-CoV-2 & Influenza A/B Multiplex Real-Time PCR Kit
REF 510010 Specification 96 Tests/Box Detection principle PCR Specimens Nasal / Nasopharyngeal swab / Oropharyngeal swab Intended Use StrongStep® SARS-CoV-2 & Influenza A/B Multiplex Real-Time PCR Kit is intended for simultaneous qualitative detection and differentiation of SARS-CoV-2,Influenza A virus and Influenza B virus RNA in healthcare provider-collected nasal and nasopharyngeal swab or oropharyngeal swab specimens and self-collected nasal or oropharyngeal swab specimens(collected in a healthcare setting with instruction by a healthcare provider) from individuals suspected of respiratory viral infection consistent with COVID-19 by their healthcare provider.
The kit is intended for use by laboratory trained personnel
-

SARS-CoV-2 IgM/IgG Antibody Rapid Test
REF 502090 Specification 20 Tests/Box Detection principle Immunochromatographic assay Specimens Whole Blood / Serum / Plasma Intended Use This is a rapid immuno-chromatographic assay for the simultaneous detection of IgM and IgG antibodies to SARS-CoV-2 virus in human whole blood, serum or plasma. The test is limited in the US to distribution to laboratories certified by CLIA to perform high complexity testing.
This test has not been reviewed by the FDA.
Negative results do not preclude acute SARS-CoV-2 infection.
Results from antibody testing should not be used to diagnose or exclude acute SARS-CoV-2 infection.
Positive results may be due to past or present infection with non-SARS-CoV-2 coronavirus strains, such as coronavirus HKU1, NL63, OC43, or 229E.







